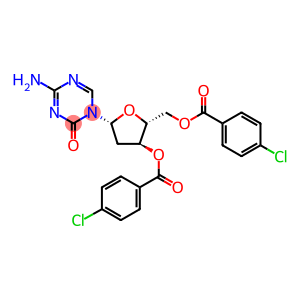
Decitabine Intermediate CR
3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine
CAS: 1034301-08-0
Molecular Formula: C22H18Cl2N4O6
Decitabine Intermediate CR - Names and Identifiers
| Name | 3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine |
| Synonyms | Decitabine Impurity 36 Decitabine Intermediate CR Decitabine Related Compound B 3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine 3',5'-Bis-O-(4-chlorobenzoyl)-2-deoxy-5-azacytosine 4-Amino-1-[3,5-bis-O-(4-chlorobenzoyl)-2-deoxy-beta-D-ribofuranosyl]-1,3,5-triazin-2(1H)-one 1,3,5-Triazin-2(1H)-one,4-amino-1-[3,5-bis-O-(4-chlorobenzoyl)-2-deoxy-b-D-erythro-pentofuranosyl]- 3',5'-Bis-O-(4-chlorobenzoyl)-2-deoxy-5-azacytosine 4-Amino-1-[3,5-bis-O-(4-chlorobenzoyl)-2-deoxy-beta-D-ribofuranosyl]-1,3,5-triazin-2(1H)-one |
| CAS | 1034301-08-0 |
| EINECS | 816-382-4 |
Decitabine Intermediate CR - Physico-chemical Properties
| Molecular Formula | C22H18Cl2N4O6 |
| Molar Mass | 505.31 |
| Density | 1.58 |
| Melting Point | >182°C (dec.) |
| Boling Point | 642.6±65.0 °C(Predicted) |
| Solubility | Chloroform (Very Slightly, Heated), DMSO (Slightly), Methanol (Slightly, Heated) |
| Appearance | Solid |
| Color | White |
| pKa | 3.38±0.20(Predicted) |
| Storage Condition | Hygroscopic, -20°C Freezer, Under inert atmosphere |
Decitabine Intermediate CR - Introduction
3 ',5'-di-p-chlorobenzoyloxy-2-deoxy-5-azacytidine, also known as ddACT, is a nucleoside compound.
Properties: ddACT is a white crystalline solid, chemically a dimer, composed of two molecules. It is highly soluble in water, but often precipitates in organic solvents. It belongs to the amide compound, has a strong intramolecular hydrogen bonding force, has a strong affinity for water molecules.
Uses: ddACT is mainly used for nucleic acid sequence analysis and drug development. It is a special nucleoside analog, mainly used in DNA sequencing technology, can be used as a substrate for chain termination reaction, used to label the end of DNA. In addition, ddACT can also be used to synthesize drugs with antiviral activity.
Production Method: ddACT can be obtained by chemical synthesis. A common synthesis method is to react 2-deoxycytidine with 3 ',5'-di-p-chlorobenzaldehyde to obtain 3 ',5'-di-chlorobenzoyloxy-2-deoxycytidine, and then Under alkaline conditions, the 3 ',5'-di-chlorobenzoyloxy-2-deoxycytidine is reacted with arylamine to further transform it into 3 ',5'-Di-p-chlorobenzoyloxy-2-deoxy-5-azacytidine.
Safety information: Due to the strong chemical properties of ddACT, it should follow safe operating procedures during operation. It may be irritating to the human skin, eyes and respiratory tract, so wear appropriate protective equipment when working to avoid direct contact. During storage and handling, it should be properly preserved and avoid contact with oxidants and other substances.
Properties: ddACT is a white crystalline solid, chemically a dimer, composed of two molecules. It is highly soluble in water, but often precipitates in organic solvents. It belongs to the amide compound, has a strong intramolecular hydrogen bonding force, has a strong affinity for water molecules.
Uses: ddACT is mainly used for nucleic acid sequence analysis and drug development. It is a special nucleoside analog, mainly used in DNA sequencing technology, can be used as a substrate for chain termination reaction, used to label the end of DNA. In addition, ddACT can also be used to synthesize drugs with antiviral activity.
Production Method: ddACT can be obtained by chemical synthesis. A common synthesis method is to react 2-deoxycytidine with 3 ',5'-di-p-chlorobenzaldehyde to obtain 3 ',5'-di-chlorobenzoyloxy-2-deoxycytidine, and then Under alkaline conditions, the 3 ',5'-di-chlorobenzoyloxy-2-deoxycytidine is reacted with arylamine to further transform it into 3 ',5'-Di-p-chlorobenzoyloxy-2-deoxy-5-azacytidine.
Safety information: Due to the strong chemical properties of ddACT, it should follow safe operating procedures during operation. It may be irritating to the human skin, eyes and respiratory tract, so wear appropriate protective equipment when working to avoid direct contact. During storage and handling, it should be properly preserved and avoid contact with oxidants and other substances.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: Decitabine Impurity 2(Decitabine USP RC B) Visit Supplier Webpage Request for quotationCAS: 1034301-08-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine Request for quotation
CAS: 1034301-08-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1034301-08-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine Visit Supplier Webpage Request for quotation
CAS: 1034301-08-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1034301-08-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Decitabine Impurity 2(Decitabine USP RC B) Visit Supplier Webpage Request for quotationCAS: 1034301-08-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine Request for quotation
CAS: 1034301-08-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1034301-08-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 3',5'-di-o-p-chlorobenzoyl-2-deoxy-5-azacytosine Visit Supplier Webpage Request for quotation
CAS: 1034301-08-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1034301-08-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


